Urea nitrate: Difference between revisions
No edit summary |
no longer a stub |
||
| (36 intermediate revisions by 26 users not shown) | |||
| Line 1: | Line 1: | ||
{{distinguish|nitrourea}} |
|||
{{chembox |
{{chembox |
||
| Verifiedfields = changed |
| Verifiedfields = changed |
||
| Watchedfields = changed |
| Watchedfields = changed |
||
| verifiedrevid = 426796341 |
| verifiedrevid = 426796341 |
||
| Name = |
|||
| ImageFile = Urea nitrate.png |
|||
| |
| ImageFile = Urea nitrate.png |
||
| ImageName = Structural formulae of the ions in urea nitrate |
|||
| ImageFile1 = Urea-nitrate-3D-balls.png |
|||
| ImageFile1 = Urea-nitrate-3D-balls.png |
|||
| ImageSize1 = 220px |
|||
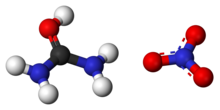
| ImageName1 = Ball-and-stick models of the ions in urea nitrate |
| ImageSize1 = 220px |
||
| ImageName1 = Ball-and-stick models of the ions in urea nitrate |
|||
| IUPACName = |
| IUPACName = |
||
| OtherNames = |
| OtherNames = |
||
| SystematicName = |
|||
|Section1={{Chembox Identifiers |
|||
| Section1 = {{Chembox Identifiers |
|||
| CASNo_Ref = {{cascite|changed|??}} |
|||
| CASNo_Ref = {{cascite|correct|CAS}} |
|||
| CASNo = 124-47-0 |
| CASNo = 124-47-0 |
||
| UNII_Ref = {{fdacite|correct|FDA}} |
|||
| UNII = DHJ35702MG |
|||
| PubChem = 31295 |
| PubChem = 31295 |
||
| ChemSpiderID_Ref = {{chemspidercite|changed|chemspider}} |
| ChemSpiderID_Ref = {{chemspidercite|changed|chemspider}} |
||
| Line 24: | Line 29: | ||
| StdInChIKey = AYTGUZPQPXGYFS-UHFFFAOYSA-N |
| StdInChIKey = AYTGUZPQPXGYFS-UHFFFAOYSA-N |
||
}} |
}} |
||
|Section2={{Chembox Properties |
| Section2 = {{Chembox Properties |
||
| C=1 | H=5 | N=3 | O=4 |
|||
| Formula = CH<sub>5</sub>N<sub>3</sub>O<sub>4</sub> |
|||
| MolarMass = 123.068 g/mol |
|||
| Appearance = |
| Appearance = |
||
| Density = 1. |
| Density = 1.67±0.011 g/cm<sup>3</sup><ref name="Characterization" /> |
||
| MeltingPtC = |
| MeltingPtC = 157-159 |
||
| BoilingPt = |
| BoilingPt = |
||
| Solubility = 167.2±0.5 mg/mL<ref name="Characterization">{{cite journal | doi = 10.1002/prep.201200178 | title = Synthesis and Characterization of Urea Nitrate and Nitrourea | date = 2013 | last1 = Oxley | first1 = Jimmie C. | last2 = Smith | first2 = James L. | last3 = Vadlamannati | first3 = Sravanthi | last4 = Brown | first4 = Austin C. | last5 = Zhang | first5 = Guang | last6 = Swanson | first6 = Devon S. | last7 = Canino | first7 = Jonathan | journal = Propellants, Explosives, Pyrotechnics | volume = 38 | issue = 3 | pages = 335–344 }}</ref> |
|||
| Solubility = 15 g/100 g |
|||
|Solubility1 = 14.2±0.1 mg/mL<ref name="Characterization" /> |
|||
| SolubleOther = soluble in alcohol<ref>http://cameochemicals.noaa.gov/chemical/12966</ref>}} |
|||
|Solvent1 = Ethanol |
|||
|Section3={{Chembox Hazards |
|||
|Solubility2 = 10.4±0.2 mg/mL<ref name="Characterization" /> |
|||
| MainHazards = |
|||
| |
|Solvent2 = Acetone |
||
|Solubility3 = 54.8±0.9 mg/mL<ref name="Characterization" /> |
|||
| AutoignitionPt =In the name of Allah the most beneficient |
|||
|Solvent3 = Methanol |
|||
the merciful. |
|||
| SolubleOther = }} |
|||
| Section3 = |
|||
Some Chemical Equations |
|||
| Section4 = {{Chembox Explosive |
|||
Introduction |
|||
| ShockSens = Low |
|||
Here are some equations: |
|||
| FrictionSens = Low |
|||
Equations |
|||
| DetonationV = 4700 m/s |
|||
eq. no Equations |
|||
| REFactor = }} |
|||
2H2+O2→2H2O |
|||
| Section5 = {{Chembox Hazards |
|||
C+O2→CO2 |
|||
| GHSPictograms = {{GHS01}} {{GHS03}} {{GHS05}} |
|||
CuSO4+Fe→FeSO4+Cu |
|||
| GHSSignalWord = '''DANGER''' |
|||
AlCl3+Fe→FeCl3+Al |
|||
| HPhrases = {{H-phrases|201|271|301|304|314|332}} |
|||
CaCl2+Cl2+C→CCl4+Ca |
|||
| PPhrases = {{P-phrases|220|233|260|250|305+351+338}} |
|||
4Na+O2→2Na2O |
|||
| NFPA-F = 1 |
|||
Fe+S→FeS |
|||
| NFPA-H = 2 |
|||
2Fe+O2→2FeO |
|||
| NFPA-R = 3 |
|||
2Zn+O2→2ZnO |
|||
CaO+CO2→CaCO3 |
|||
H2+I2→2HI |
|||
H2+F2→2HF |
|||
H2+Cl2→2HCl |
|||
H2+Br2→2HBr |
|||
2Pb+O2→2PbO |
|||
SiO2+4Na→Na2O+Si |
|||
2H2+C→CH4 |
|||
N2+3H2→2NH3 |
|||
2C+H2→C2H2 |
|||
C2H5OH→H2O+C2H4 |
|||
CO2+SO2→2O2+C+S |
|||
Cs2(SO4)3+3H2→3H2SO4+2Cs |
|||
2NaOH+H2+Cl2→2NaCl+2H2O |
|||
H2S+Fe→FeS+H2 |
|||
CaCl2+2Na→Ca+2NaCl |
|||
CaSO4+Cu→CuSO4+Ca |
|||
Reaction of Acid with metal oxide |
|||
Acids react with metal oxides to form salt and water. There are some of such equations: |
|||
27. FeO+H2SO4→FeSO4+H2O |
|||
28. ZnO+2HCl→ZnCl2+H2O |
|||
29. CuO+2HNO3→H2O+Cu(NO3)2 |
|||
30. PbO+2HNO3→Pb(NO3)2+H2O |
|||
31. Na2O+2HNO3→H2O+2NaNO3 |
|||
32. FeO+2HNO3→Fe(NO3)2+H2O |
|||
33. ZnO+2HNO3→H2O+Zn(NO3)2 |
|||
34. FeO+2HCl→FeCl2+H2O |
|||
35. ZnO+H2SO4→ZnSO4+H2O |
|||
36. CuO+2HCl→CuCl2+H2O |
|||
37. PbO+2HCl→PbCl2+H2O |
|||
38. Na2O+2HCl→2NaCl+H2O |
|||
39. CaO+2HCl→CaCl2+H2O |
|||
40. MgO+2HCl→MgCl2+H2O |
|||
41. CuO+H2SO4→CuSO4 |
|||
42. PbO+H2SO4→PbSO4+H2O |
|||
43. Na2O+H2SO4→Na2SO4+H2O |
|||
44. CaO+H2SO4→CaSO4+H2O |
|||
45. MgO+H2SO4→MgSO4+H2O |
|||
Reaction of Acid with metal |
|||
Acids react with metals to give salt and water. Some chemical equations are given below: |
|||
46. H2SO4+Cu→CuSO4+H2 |
|||
47. H2SO4+Zn→ZnSO4+H2 |
|||
48. 2HCl+Ca→CaCl2+H2 |
|||
49. 2HCl+Mg→MgCl2+H2 |
|||
50. Fe+2HNO3→Fe(NO3)2+H2O |
|||
Reaction to prepare ozone artfically |
|||
Scientists tell us that the formula of ozone is O3. This layer protects the Earth from radioactive rays (which are alpha, beta and gamma rays). The equation to prepare ozone artifially: |
|||
2O2+O2→2O3 |
|||
Reaction to estinguish fire |
|||
In the burning fire process it happens by reaction of oxygen gas (O2) and carbon/sulphur. To lit fire we first have to remove carbon/sulphur. Here is an equation to lit fire: |
|||
CO2+2H2→2H2O+C |
|||
Or |
|||
SO2+2Ca→2CaO+S |
|||
Redox reactions to remove greenhouse gases |
|||
Reactions in which oxidation state or oxidation number (a partial charge assigned to a substance) changes are called oxidation-reduction or redox reactions. |
|||
When rays of the Sun reach the Earth's atmosphere, some of them hit dust particles, and some are reflected by them. Some hit the clouds are absorbed by them. Some gases help the rays that not go back to space. The gases are called greenhouse gases. This whole process (excluding the definition of redox reactions) is called Greenhouse effect. Carbondioxide (CO2), Methane (CH4) and Nitrous oxide (N2O) are some important greenhouse gases. I will tell you how to make these gases useful. Here are equations to make these gases harmless: |
|||
N2O+2O2+H2O+2CaO→2CaNO3+H2+O2 |
|||
CH4+C+3O2+CaO→2CaCO3+H2O |
|||
CO2+H2O+NaOH→H2O+NaHCO3 |
|||
}} |
}} |
||
| Section6 = |
|||
}} |
}} |
||
[[File:Urea nitrate (IV).jpg|thumb|Crystals of urea nitrate]] |
|||
'''Urea nitrate''' is a fertilizer-based [[high explosive]] that has been used in [[improvised explosive device]]s in [[Afghanistan]], [[Pakistan]], [[Iraq]], and various other [[terrorism|terrorist]] acts elsewhere in the world, like the [[1993 World Trade Center bombings]].<ref name="wired">{{cite news| |
|||
'''Urea nitrate''' is a fertilizer-based [[high explosive]] that has been used in [[improvised explosive device]]s in [[Afghanistan]], [[Pakistan]], [[Iraq]], and various [[terrorism|terrorist]] acts elsewhere in the world such as in the [[1993 World Trade Center bombings]].<ref name="wired">{{cite news| |
|||
url=http://blog.wired.com/wiredscience/2007/09/spray-on-test-f.html| |
url=http://blog.wired.com/wiredscience/2007/09/spray-on-test-f.html| |
||
title=Chem Lab: Spray-On Test for Improvised Explosives| |
title=Chem Lab: Spray-On Test for Improvised Explosives| |
||
author=Aaron Rowe| |
author=Aaron Rowe| |
||
publisher=Wired |
publisher=Wired |
||
| date =18 September 2007}}</ref> It has a destructive power similar to better-known [[ammonium nitrate]] explosives, with a [[velocity of detonation]] between {{convert|11155|ft/s|m/s|abbr=on}} and {{convert|15420|ft/s|m/s|abbr=on}}.<ref name="globalsecurity">{{cite web| |
| date =18 September 2007}}</ref> It has a destructive power similar to better-known [[ammonium nitrate]] explosives, with a [[velocity of detonation]] between {{convert|11155|ft/s|m/s|abbr=on|order=flip}} and {{convert|15420|ft/s|m/s|abbr=on|order=flip}}.<ref name="globalsecurity">{{cite web| |
||
url=http://www.globalsecurity.org/military/systems/munitions/explosives-anfo.htm| |
url=http://www.globalsecurity.org/military/systems/munitions/explosives-anfo.htm| |
||
title=Explosives - ANFO (Ammonium Nitrate - Fuel Oil)| |
title=Explosives - ANFO (Ammonium Nitrate - Fuel Oil)| |
||
publisher=GlobalSecurity.org}}</ref> |
publisher=GlobalSecurity.org}}</ref> It has chemical formula of {{chem2|CH5N3O4}} or {{chem2|(NH2)2COHNO3}}. |
||
Urea nitrate is produced in one step by reaction of [[urea]] with [[nitric acid]]. This is an [[exothermic reaction]], so steps must be taken to control the temperature. |
Urea nitrate is produced in one step by reaction of [[urea]] with [[nitric acid]]. This is an [[exothermic reaction]], so steps must be taken to control the temperature. |
||
Urea nitrate explosions may be initiated using a blasting cap.<ref name="globalsecurity" /> |
Urea nitrate explosions may be initiated using a [[blasting cap]].<ref name="globalsecurity" /> |
||
==Chemistry== |
==Chemistry== |
||
Urea contains a [[Carbonyl|carbonyl group]]. The more electronegative oxygen atom pulls electrons away from the carbon forming a greater electron density around the oxygen, giving |
Urea contains a [[Carbonyl|carbonyl group]]. The more electronegative oxygen atom pulls electrons away from the carbon atom, forming a polar bond with greater electron density around the oxygen atom, giving it a partial negative charge. In a simplistic sense, nitric acid dissociates in aqueous solution into protons (hydrogen cations) and nitrate anions. The electrophilic proton contributed by the acid is attracted to the negatively charged oxygen atom on the urea molecule and the two form a covalent bond. The formed O-H bond is stabilized into a hydroxyl group when the oxygen abstracts an electron pair away from the central carbon atom, which leads to bond resonance between it and the two amino groups. As such, the urea cation can be thought of as a amidinium species. Paired with the spectator nitrate counteranion, it forms urea nitrate. |
||
:{{chem2 | (NH2)2CO (aq) + HNO3 (aq) -> [(NH2)2COH]+[NO3]− (s) }} |
|||
(NH<sub>2</sub>)<sub>2</sub>CO (aq) + HNO<sub>3</sub> (aq) → (NH<sub>2</sub>)<sub>2</sub>COHNO<sub>3</sub> (s) |
|||
The compound is favored by many amateur explosive enthusiasts as a principal explosive for use in larger charges. In this role it acts as a substitute for ammonium nitrate based explosives. This is due to the ease of acquiring the materials necessary to synthesize it, and its greater sensitivity to initiation compared to ammonium nitrate based explosives. |
The compound is favored by many amateur explosive enthusiasts as a principal explosive for use in larger charges. In this role it acts as a substitute for ammonium nitrate based explosives. This is due to the ease of acquiring the materials necessary to synthesize it, and its greater sensitivity to initiation compared to ammonium nitrate based explosives. |
||
| Line 139: | Line 86: | ||
==Further reading== |
==Further reading== |
||
* |
*{{cite journal |vauthors=Almog J, Burda G, Shloosh Y, Abramovich-Bar S, Wolf E, Tamiri T |title=Recovery and detection of urea nitrate in traces |journal=J. Forensic Sci. |volume=52 |issue=6 |pages=1284–90 |date=November 2007 |pmid=17868267 |doi=10.1111/j.1556-4029.2007.00551.x |s2cid=10228717 }} |
||
* |
*{{cite journal |author=Mr.X |title=Improvised Urea Nitrate |journal=Aware EZine Gamma |date=July 2008 |url=http://www.awarenetwork.org/etc/gamma/?x=5 |access-date=2008-11-11 |archive-url=https://web.archive.org/web/20080920082234/http://www.awarenetwork.org/etc/gamma/?x=5 |archive-date=2008-09-20 |url-status=dead }} |
||
==External links== |
|||
[[File:Urea nitrate (IV).jpg|thumb|Urea nitrate]] |
|||
*{{Commonscatinline}} |
|||
[[Category:Explosive chemicals]] |
[[Category:Explosive chemicals]] |
||
[[Category:Nitrates]] |
[[Category:Nitrates]] |
||
[[Category:Ureas]] |
[[Category:Ureas]] |
||
{{explosive-stub}} |
|||
Latest revision as of 18:13, 3 April 2024

| |
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.004.276 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| CH5N3O4 | |
| Molar mass | 123.068 g·mol−1 |
| Density | 1.67±0.011 g/cm3[1] |
| Melting point | 157–159 °C (315–318 °F; 430–432 K) |
| 167.2±0.5 mg/mL[1] | |
| Solubility in Ethanol | 14.2±0.1 mg/mL[1] |
| Solubility in Acetone | 10.4±0.2 mg/mL[1] |
| Solubility in Methanol | 54.8±0.9 mg/mL[1] |
| Explosive data | |
| Shock sensitivity | Low |
| Friction sensitivity | Low |
| Detonation velocity | 4700 m/s |
| Hazards | |
| GHS labelling: | |
  
| |
| Danger | |
| H201, H271, H301, H304, H314, H332 | |
| P220, P233, P250, P260, P305+P351+P338 | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |

Urea nitrate is a fertilizer-based high explosive that has been used in improvised explosive devices in Afghanistan, Pakistan, Iraq, and various terrorist acts elsewhere in the world such as in the 1993 World Trade Center bombings.[2] It has a destructive power similar to better-known ammonium nitrate explosives, with a velocity of detonation between 3,400 m/s (11,155 ft/s) and 4,700 m/s (15,420 ft/s).[3] It has chemical formula of CH5N3O4 or (NH2)2COHNO3.
Urea nitrate is produced in one step by reaction of urea with nitric acid. This is an exothermic reaction, so steps must be taken to control the temperature.
Urea nitrate explosions may be initiated using a blasting cap.[3]
Chemistry
[edit]Urea contains a carbonyl group. The more electronegative oxygen atom pulls electrons away from the carbon atom, forming a polar bond with greater electron density around the oxygen atom, giving it a partial negative charge. In a simplistic sense, nitric acid dissociates in aqueous solution into protons (hydrogen cations) and nitrate anions. The electrophilic proton contributed by the acid is attracted to the negatively charged oxygen atom on the urea molecule and the two form a covalent bond. The formed O-H bond is stabilized into a hydroxyl group when the oxygen abstracts an electron pair away from the central carbon atom, which leads to bond resonance between it and the two amino groups. As such, the urea cation can be thought of as a amidinium species. Paired with the spectator nitrate counteranion, it forms urea nitrate.
- (NH2)2CO (aq) + HNO3 (aq) → [(NH2)2COH]+[NO3]− (s)
The compound is favored by many amateur explosive enthusiasts as a principal explosive for use in larger charges. In this role it acts as a substitute for ammonium nitrate based explosives. This is due to the ease of acquiring the materials necessary to synthesize it, and its greater sensitivity to initiation compared to ammonium nitrate based explosives.
References
[edit]- ^ a b c d e Oxley, Jimmie C.; Smith, James L.; Vadlamannati, Sravanthi; Brown, Austin C.; Zhang, Guang; Swanson, Devon S.; Canino, Jonathan (2013). "Synthesis and Characterization of Urea Nitrate and Nitrourea". Propellants, Explosives, Pyrotechnics. 38 (3): 335–344. doi:10.1002/prep.201200178.
- ^ Aaron Rowe (18 September 2007). "Chem Lab: Spray-On Test for Improvised Explosives". Wired.
- ^ a b "Explosives - ANFO (Ammonium Nitrate - Fuel Oil)". GlobalSecurity.org.
Further reading
[edit]- Almog J, Burda G, Shloosh Y, Abramovich-Bar S, Wolf E, Tamiri T (November 2007). "Recovery and detection of urea nitrate in traces". J. Forensic Sci. 52 (6): 1284–90. doi:10.1111/j.1556-4029.2007.00551.x. PMID 17868267. S2CID 10228717.
- Mr.X (July 2008). "Improvised Urea Nitrate". Aware EZine Gamma. Archived from the original on 2008-09-20. Retrieved 2008-11-11.
External links
[edit] Media related to Urea nitrate at Wikimedia Commons
Media related to Urea nitrate at Wikimedia Commons

