Acetoacetic acid: Difference between revisions
identical refs consolidated |
No edit summary |
||
| (28 intermediate revisions by 15 users not shown) | |||
| Line 7: | Line 7: | ||
| ImageFile1 = Acetoacetic-acid-3D-balls.png |
| ImageFile1 = Acetoacetic-acid-3D-balls.png |
||
| PIN = 3-Oxobutanoic acid<ref name=iupac2013>{{cite book | title = Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book) | publisher = [[Royal Society of Chemistry|The Royal Society of Chemistry]] | date = 2014 | location = Cambridge | page = 748 | doi = 10.1039/9781849733069-FP001 | isbn = 978-0-85404-182-4| chapter = Front Matter }}</ref> |
| PIN = 3-Oxobutanoic acid<ref name=iupac2013>{{cite book | title = Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book) | publisher = [[Royal Society of Chemistry|The Royal Society of Chemistry]] | date = 2014 | location = Cambridge | page = 748 | doi = 10.1039/9781849733069-FP001 | isbn = 978-0-85404-182-4| chapter = Front Matter }}</ref> |
||
| |
| SystematicName = 3-Oxobutyric acid |
||
| OtherNames = Acetoacetic acid<br/>Diacetic acid<br/>Acetylacetic acid<br/>Acetonecarboxylic acid |
|||
|Section1={{Chembox Identifiers |
|Section1={{Chembox Identifiers |
||
| KEGG_Ref = {{keggcite|correct|kegg}} |
| KEGG_Ref = {{keggcite|correct|kegg}} |
||
| Line 22: | Line 23: | ||
| CASNo_Ref = {{cascite|correct|CAS}} |
| CASNo_Ref = {{cascite|correct|CAS}} |
||
| CASNo = 541-50-4 |
| CASNo = 541-50-4 |
||
| UNII_Ref = {{fdacite|correct|FDA}} |
|||
| UNII = 4ZI204Y1MC |
|||
| ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} |
| ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} |
||
| ChemSpiderID = 94 |
| ChemSpiderID = 94 |
||
| Line 32: | Line 35: | ||
|Section2={{Chembox Properties |
|Section2={{Chembox Properties |
||
| C=4 | H=6 | O=3 |
| C=4 | H=6 | O=3 |
||
| ⚫ | |||
| MolarMass = 102.088 g/mol |
|||
| ⚫ | |||
| Density = |
| Density = |
||
| MeltingPtC = 36.5 |
| MeltingPtC = 36.5 |
||
| BoilingPt = Decomposes |
| BoilingPt = Decomposes |
||
| Solubility = |
| Solubility = Soluble |
||
| SolubleOther = |
| SolubleOther = Soluble in [[ethanol]], [[diethyl ether|ether]] |
||
| Solvent = organic solvents |
|||
| pKa = 3.58 |
| pKa = 3.58<ref>Dawson, R. M. C., et al., ''Data for Biochemical Research'', Oxford, Clarendon Press, 1959.</ref> |
||
}} |
}} |
||
}} |
}} |
||
'''Acetoacetic acid''' (also |
'''Acetoacetic acid''' ([[IUPAC name]]: '''3-Oxobutanoic acid''', also known as '''Acetonecarboxylic acid''' or '''Diacetic acid''') is the [[organic compound]] with the formula CH<sub><span style="font-size:100%;">3</span></sub>COCH<sub><span style="font-size:100%;">2</span></sub>COOH. It is the simplest beta-[[keto acid]], and like other members of this class, it is unstable. The [[Methyl acetoacetate|methyl]] and [[Ethyl acetoacetate|ethyl]] esters, which are quite stable, are produced on a large scale industrially as precursors to dyes. Acetoacetic acid is a [[weak acid]].<ref name=Ullmann/> |
||
==Biochemistry== |
==Biochemistry== |
||
Under typical physiological conditions, acetoacetic acid exists as its [[conjugate base]], '''acetoacetate''' |
Under typical physiological conditions, acetoacetic acid exists as its [[conjugate base]], '''acetoacetate''': |
||
:{{chem2|[[Acetyl|Ac]]CH2CO2H -> AcCH2CO2- + H+}} |
|||
Unbound acetoacetate is primarily produced by [[liver]] [[mitochondria]] from its [[thioester]] with [[coenzyme A]] (CoA): |
|||
:{{chem2|AcCH2C(O)\sCoA + OH- -> AcCH2CO2- + H\sCoA}} |
|||
The acetoacetate-CoA itself is formed by three routes: |
|||
*[[HMG-CoA|3-hydroxy-3-methylglutaryl CoA]] releases acetyl CoA and acetoacetate: |
|||
*:{{chem2|-O2CCH2\sC([[Methyl|Me]])(OH)\sCH2C(O)\sCoA -> -O2CCH2\sAc + Ac\sCoA}} |
|||
*Acetoacetyl-CoA can come from [[beta oxidation]] of [[butyryl-CoA]]: |
|||
*:{{chem2|[[Et]]\sCH2C(O)\sCoA + 2NAD+ + H2O + FAD -> Ac\sCH2C(O)\sCoA + 2NADH + FADH2}} |
|||
*Condensation of pair of acetyl CoA molecules as catalyzed by [[thiolase]].<ref name=Stryer1981>{{cite book|author=Lubert Stryer|author-link=Lubert Stryer|title=Biochemistry|date=1981 |edition=2nd|title-link=Biochemistry (Stryer)}}</ref>{{rp|393}} |
|||
*:{{chem2|2Ac\sCoA -> AcCH2C(O)\sCoA + H\sCoA}} |
|||
| ⚫ | In mammals, acetoacetate produced in the liver (along with the other two "[[ketone bodies]]") is released into the bloodstream as an energy source during periods of [[fasting]], exercise, or as a result of [[type 1 diabetes mellitus]].<ref name=stryer2>{{cite book |last1= Stryer |first1= Lubert | title=Biochemistry. | edition= Fourth |location= New York |publisher= W.H. Freeman and Company|date= 1995 |pages= 510–515, 581–613, 775–778 |isbn= 0-7167-2009-4 }}</ref> First, a CoA group is enzymatically transferred to it from [[succinyl CoA]], converting it back to acetoacetyl CoA; this is then broken into two acetyl CoA molecules by thiolase, and these then enter the [[citric acid cycle]]. Heart muscle and renal cortex prefer acetoacetate over glucose. The brain uses acetoacetate when glucose levels are low due to fasting or diabetes.<ref name=Stryer1981/>{{rp|394}} |
||
Acetoacetate is produced in the [[mitochondria]] of the [[liver]] from acetoacetyl [[coenzyme A]] (CoA). First, another [[acetyl]] group is added from [[acetyl CoA]] to form [[HMG-CoA|3-hydroxy-3-methylgluteryl CoA]], then an acetyl CoA is lost from this, yielding acetoacetate. The initial acetoacetate can come from the last cycle in the [[beta oxidation]] of a [[fatty acid]], or it can be synthesized from two acetyl CoA molecules, catalyzed by [[thiolase]].<ref name=Stryer1981>{{cite book|author=Lubert Stryer|author-link=Lubert Stryer|title=Biochemistry|date=1981 |edition=2nd|title-link=Biochemistry (Stryer)}}</ref>{{rp|393}} |
|||
| ⚫ | In mammals, acetoacetate produced in the liver (along with the other two "[[ketone bodies]]") is released into the bloodstream as an energy source during periods of [[fasting]], exercise, or as a result of [[type 1 diabetes mellitus]].<ref name=stryer2>{{cite book |last1= Stryer |first1= Lubert | title=Biochemistry. | edition= Fourth |location= New York |publisher= W.H. Freeman and Company|date= 1995 |pages= 510–515, 581–613, 775–778 |isbn= 0 |
||
== Synthesis and properties == |
== Synthesis and properties == |
||
Acetoacetic acid may be prepared by the hydrolysis of [[diketene]]. Its esters are produced analogously via |
Acetoacetic acid may be prepared by the hydrolysis of [[diketene]]. Its esters are produced analogously via reactions between diketene and alcohols,<ref name=Ullmann>{{cite book|author=Franz Dietrich Klingler|author2=Wolfgang Ebertz|chapter=Oxocarboxylic Acids|title=Ullmann's Encyclopedia of Industrial Chemistry|year=2005|publisher=Wiley-VCH|place=Weinheim|doi=10.1002/14356007.a18_313|isbn=978-3527306732}}</ref> and acetoacetic acid can be prepared by the hydrolysis of these species.<ref>{{cite journal |
||
| author = Robert C. Krueger |
| author = Robert C. Krueger |
||
| title = Crystalline Acetoacetic Acid |
| title = Crystalline Acetoacetic Acid |
||
| Line 63: | Line 74: | ||
| doi = 10.1021/ja01141a521 }} |
| doi = 10.1021/ja01141a521 }} |
||
</ref> |
</ref> |
||
In general, acetoacetic acid is generated at 0 °C and used in situ immediately.<ref>{{OrgSynth|last1=Reynolds|first1=George A.|last2=VanAllan|first2=J. A.|title=Methylglyoxal-ω-Phenylhydrazone|journal=Organic Syntheses|year=1952|volume=32|pages=84|prep=CV4P0633|collvol=4|collvolpages=633|doi=10.15227/orgsyn.032.0084}}</ref> |
In general, acetoacetic acid is generated at 0 °C and used in situ immediately.<ref>{{OrgSynth|last1=Reynolds|first1=George A.|last2=VanAllan|first2=J. A.|title=Methylglyoxal-ω-Phenylhydrazone|journal=Organic Syntheses|year=1952|volume=32|pages=84|prep=CV4P0633|collvol=4|collvolpages=633|doi=10.15227/orgsyn.032.0084}}</ref> |
||
It decomposes at a moderate [[reaction rate]] into [[acetone]] and [[carbon dioxide]]: |
|||
:{{chem2|CH3C(O)CH2CO2H → CH3C(O)CH3 + CO2}} |
|||
:CH<sub>3</sub>C(O)CH<sub>2</sub>CO<sub>2</sub>H → CH<sub>3</sub>C(O)CH<sub>3</sub> + CO<sub>2</sub> |
|||
The acid form has a [[half-life]] of 140 minutes at 37 °C in water, whereas the basic form (the anion) has a half-life of 130 hours. That is, it reacts about 55 times more slowly.<ref>{{cite journal |doi= 10.1071/CH9671823 |author1=Hay, R. W. |author2=Bond, M. A. |year= 1967|title= Kinetics of decarboxilation of acetoacetic acid|journal= Aust. J. Chem.|volume = 20|issue=9|pages=1823–8}}</ref> |
The acid form has a [[half-life]] of 140 minutes at 37 °C in water, whereas the basic form (the anion) has a half-life of 130 hours. That is, it reacts about 55 times more slowly.<ref>{{cite journal |doi= 10.1071/CH9671823 |author1=Hay, R. W. |author2=Bond, M. A. |year= 1967|title= Kinetics of decarboxilation of acetoacetic acid|journal= Aust. J. Chem.|volume = 20|issue=9|pages=1823–8}}</ref> The corresponding decarboxylation of trifluoroacetoacetate is used to prepare [[trifluoroacetone]]: |
||
:{{chem2|CF3C(O)CH2CO2H → CF3C(O)CH3 + CO2}} |
|||
| ⚫ | |||
| ⚫ | |||
Acetoacetic acid displays keto-enol [[tautomerisation]], with the enol form being partially stabilised by extended conjugation and intramolecular [[H-bonding]]. The equilibrium is strongly solvent depended; with the keto form dominating in polar solvents (98% in water) and the enol form accounting for 25-49% of material in non-polar solvents.<ref>{{cite journal|last1=Grande|first1=Karen D.|last2=Rosenfeld|first2=Stuart M.|title=Tautomeric equilibriums in acetoacetic acid|journal=The Journal of Organic Chemistry|volume=45|issue=9|year=1980|pages=1626–1628|issn=0022-3263|doi=10.1021/jo01297a017}}</ref> |
Acetoacetic acid displays keto-enol [[tautomerisation]], with the enol form being partially stabilised by extended conjugation and intramolecular [[H-bonding]]. The equilibrium is strongly solvent depended; with the keto form dominating in polar solvents (98% in water) and the enol form accounting for 25-49% of material in non-polar solvents.<ref>{{cite journal|last1=Grande|first1=Karen D.|last2=Rosenfeld|first2=Stuart M.|title=Tautomeric equilibriums in acetoacetic acid|journal=The Journal of Organic Chemistry|volume=45|issue=9|year=1980|pages=1626–1628|issn=0022-3263|doi=10.1021/jo01297a017}}</ref> |
||
| Line 73: | Line 88: | ||
==Applications== |
==Applications== |
||
| ⚫ | |||
| ⚫ | Acetoacetic esters are used for the [[acetoacetylation]] reaction, which is widely used in the production of [[arylide yellow]]s and [[diarylide]] dyes.<ref name=Ullmann/> Although the esters can be used in this reaction, diketene also reacts with [[alcohol]]s and [[amine]]s to the corresponding acetoacetic acid derivatives in a process called '''acetoacetylation'''. An example is the reaction with 4-amino[[indane]]:<ref>{{cite journal | title = Knorr Cyclizations and Distonic Superelectrophiles |author1=Kiran Kumar Solingapuram Sai |author2=Thomas M. Gilbert |author3=Douglas A. Klumpp | journal = [[J. Org. Chem.]] | year = 2007 | volume = 72 | pages = 9761–9764 | doi = 10.1021/jo7013092 | pmid = 17999519 | issue = 25}}</ref> |
||
| ⚫ | Acetoacetic esters are used for the [[acetoacetylation]] reaction, which is widely used in the production of [[arylide yellow]]s and [[diarylide]] dyes.<ref name=Ullmann/> Although the esters can be used in this reaction, diketene also reacts with [[Alcohol (chemistry)|alcohol]]s and [[amine]]s to the corresponding acetoacetic acid derivatives in a process called '''acetoacetylation'''. An example is the reaction with 4-amino[[indane]]:<ref>{{cite journal | title = Knorr Cyclizations and Distonic Superelectrophiles |author1=Kiran Kumar Solingapuram Sai |author2=Thomas M. Gilbert |author3=Douglas A. Klumpp | journal = [[J. Org. Chem.]] | year = 2007 | volume = 72 | pages = 9761–9764 | doi = 10.1021/jo7013092 | pmid = 17999519 | issue = 25}}</ref> |
||
[[Image:DiketeneReaction.svg|400px|Diketene reaction Sai 2007]] |
|||
| ⚫ | |||
== Detection == |
== Detection == |
||
Acetoacetic acid is measured in the urine of people with diabetes to test for [[Ketone bodies#Ketosis and ketoacidosis|ketoacidosis]]<ref name=Nyenwe2016/> and for monitoring people on a [[ketogenic diet|ketogenic]] or [[low-carbohydrate diet]] |
Acetoacetic acid is measured in the urine of people with diabetes to test for [[Ketone bodies#Ketosis and ketoacidosis|ketoacidosis]]<ref name=Nyenwe2016/> and for monitoring people on a [[ketogenic diet|ketogenic]] or [[low-carbohydrate diet]].<ref name=Hartman2007b>{{cite journal|last1=Hartman|first1=AL|last2=Vining|first2=EP|title=Clinical aspects of the ketogenic diet|journal=Epilepsia|date=January 2007|volume=48|issue=1|pages=31–42|doi=10.1111/j.1528-1167.2007.00914.x|pmid=17241206|doi-access=|s2cid=21195842 }}</ref><ref>{{cite journal|last1=Sumithran|first1=Priya|last2=Proietto |first2=Joseph|title=Ketogenic diets for weight loss: A review of their principles, safety and efficacy|journal=Obesity Research & Clinical Practice|date=2008 |volume=2|issue=1|pages=I-II|doi=10.1016/j.orcp.2007.11.003|pmid=24351673}}</ref> This is done using dipsticks coated in [[nitroprusside]] or similar reagents. Nitroprusside changes from pink to purple in the presence of acetoacetate, the [[conjugate acid|conjugate base]] of acetoacetic acid, and the colour change is graded by eye. The test does not measure β-hydroxybutyrate, the most abundant ketone in the body; during treatment of ketoacidosis [[Beta-Hydroxybutyric acid|β-hydroxybutyrate]] is converted to acetoacetate so the test is not useful after treatment begins<ref name=Nyenwe2016>{{cite journal|last1=Nyenwe|first1=EA|last2=Kitabchi|first2=AE|title=The evolution of diabetic ketoacidosis: An update of its etiology, pathogenesis and management|journal=Metabolism: Clinical and Experimental|date=April 2016|volume=65|issue=4|pages=507–21|pmid=26975543|doi=10.1016/j.metabol.2015.12.007}}</ref> and may be falsely low at diagnosis.<ref>{{cite journal|last1=Misra|first1=S|last2=Oliver|first2=NS|title=Diabetic ketoacidosis in adults.|journal=BMJ (Clinical Research Ed.)|date=28 October 2015|volume=351|pages=h5660|pmid=26510442|doi=10.1136/bmj.h5660|url=http://spiral.imperial.ac.uk/bitstream/10044/1/41091/2/bmj.h5660.full.pdf|hdl=10044/1/41091|s2cid=2819002|hdl-access=free}}</ref> |
||
Similar tests are used in [[dairy cows]] to test for [[Ketosis#Veterinary medicine|ketosis]].<ref>{{cite journal|last1=Tatone|first1=EH|last2=Gordon|first2=JL|last3=Hubbs|first3=J|last4=LeBlanc|first4=SJ|last5=DeVries|first5=TJ|last6=Duffield|first6=TF|title=A systematic review and meta-analysis of the diagnostic accuracy of point-of-care tests for the detection of hyperketonemia in dairy cows.|journal=Preventive Veterinary Medicine|date=1 August 2016|volume=130|pages=18–32|pmid=27435643|doi=10.1016/j.prevetmed.2016.06.002}}</ref> |
Similar tests are used in [[dairy cows]] to test for [[Ketosis#Veterinary medicine|ketosis]].<ref>{{cite journal|last1=Tatone|first1=EH|last2=Gordon|first2=JL|last3=Hubbs|first3=J|last4=LeBlanc|first4=SJ|last5=DeVries|first5=TJ|last6=Duffield|first6=TF|title=A systematic review and meta-analysis of the diagnostic accuracy of point-of-care tests for the detection of hyperketonemia in dairy cows.|journal=Preventive Veterinary Medicine|date=1 August 2016|volume=130|pages=18–32|pmid=27435643|doi=10.1016/j.prevetmed.2016.06.002}}</ref> |
||
| Line 85: | Line 101: | ||
==See also== |
==See also== |
||
* [[3-Hydroxybutyrate dehydrogenase]] |
* [[3-Hydroxybutyrate dehydrogenase]] |
||
* [[Ethyl acetoacetate]] |
|||
==References== |
==References== |
||
| Line 94: | Line 111: | ||
[[Category:Histone deacetylase inhibitors]] |
[[Category:Histone deacetylase inhibitors]] |
||
[[Category:Beta-keto acids]] |
[[Category:Beta-keto acids]] |
||
[[Category:Acetoacetates| ]] |
|||
Latest revision as of 08:51, 24 August 2024

| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
3-Oxobutanoic acid[1] | |
| Systematic IUPAC name
3-Oxobutyric acid | |
| Other names
Acetoacetic acid
Diacetic acid Acetylacetic acid Acetonecarboxylic acid | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H6O3 | |
| Molar mass | 102.089 g·mol−1 |
| Appearance | Colorless, oily liquid |
| Melting point | 36.5 °C (97.7 °F; 309.6 K) |
| Boiling point | Decomposes |
| Soluble | |
| Solubility in organic solvents | Soluble in ethanol, ether |
| Acidity (pKa) | 3.58[2] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
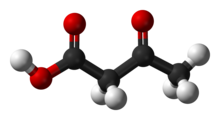
Acetoacetic acid (IUPAC name: 3-Oxobutanoic acid, also known as Acetonecarboxylic acid or Diacetic acid) is the organic compound with the formula CH3COCH2COOH. It is the simplest beta-keto acid, and like other members of this class, it is unstable. The methyl and ethyl esters, which are quite stable, are produced on a large scale industrially as precursors to dyes. Acetoacetic acid is a weak acid.[3]
Biochemistry
[edit]Under typical physiological conditions, acetoacetic acid exists as its conjugate base, acetoacetate:
- AcCH2CO2H → AcCH2CO−2 + H+
Unbound acetoacetate is primarily produced by liver mitochondria from its thioester with coenzyme A (CoA):
- AcCH2C(O)−CoA + OH− → AcCH2CO−2 + H−CoA
The acetoacetate-CoA itself is formed by three routes:
- 3-hydroxy-3-methylglutaryl CoA releases acetyl CoA and acetoacetate:
- −O2CCH2−C(Me)(OH)−CH2C(O)−CoA → −O2CCH2−Ac + Ac−CoA
- Acetoacetyl-CoA can come from beta oxidation of butyryl-CoA:
- Et−CH2C(O)−CoA + 2NAD+ + H2O + FAD → Ac−CH2C(O)−CoA + 2NADH + FADH2
- Condensation of pair of acetyl CoA molecules as catalyzed by thiolase.[4]: 393
- 2Ac−CoA → AcCH2C(O)−CoA + H−CoA
In mammals, acetoacetate produced in the liver (along with the other two "ketone bodies") is released into the bloodstream as an energy source during periods of fasting, exercise, or as a result of type 1 diabetes mellitus.[5] First, a CoA group is enzymatically transferred to it from succinyl CoA, converting it back to acetoacetyl CoA; this is then broken into two acetyl CoA molecules by thiolase, and these then enter the citric acid cycle. Heart muscle and renal cortex prefer acetoacetate over glucose. The brain uses acetoacetate when glucose levels are low due to fasting or diabetes.[4]: 394
Synthesis and properties
[edit]Acetoacetic acid may be prepared by the hydrolysis of diketene. Its esters are produced analogously via reactions between diketene and alcohols,[3] and acetoacetic acid can be prepared by the hydrolysis of these species.[6] In general, acetoacetic acid is generated at 0 °C and used in situ immediately.[7]
It decomposes at a moderate reaction rate into acetone and carbon dioxide:
- CH3C(O)CH2CO2H → CH3C(O)CH3 + CO2
The acid form has a half-life of 140 minutes at 37 °C in water, whereas the basic form (the anion) has a half-life of 130 hours. That is, it reacts about 55 times more slowly.[8] The corresponding decarboxylation of trifluoroacetoacetate is used to prepare trifluoroacetone:
- CF3C(O)CH2CO2H → CF3C(O)CH3 + CO2
It is a weak acid (like most alkyl carboxylic acids), with a pKa of 3.58.
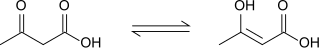
Acetoacetic acid displays keto-enol tautomerisation, with the enol form being partially stabilised by extended conjugation and intramolecular H-bonding. The equilibrium is strongly solvent depended; with the keto form dominating in polar solvents (98% in water) and the enol form accounting for 25-49% of material in non-polar solvents.[9]
Applications
[edit]
Acetoacetic esters are used for the acetoacetylation reaction, which is widely used in the production of arylide yellows and diarylide dyes.[3] Although the esters can be used in this reaction, diketene also reacts with alcohols and amines to the corresponding acetoacetic acid derivatives in a process called acetoacetylation. An example is the reaction with 4-aminoindane:[10]
Detection
[edit]Acetoacetic acid is measured in the urine of people with diabetes to test for ketoacidosis[11] and for monitoring people on a ketogenic or low-carbohydrate diet.[12][13] This is done using dipsticks coated in nitroprusside or similar reagents. Nitroprusside changes from pink to purple in the presence of acetoacetate, the conjugate base of acetoacetic acid, and the colour change is graded by eye. The test does not measure β-hydroxybutyrate, the most abundant ketone in the body; during treatment of ketoacidosis β-hydroxybutyrate is converted to acetoacetate so the test is not useful after treatment begins[11] and may be falsely low at diagnosis.[14]
Similar tests are used in dairy cows to test for ketosis.[15]
See also
[edit]References
[edit]- ^ "Front Matter". Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 748. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
- ^ Dawson, R. M. C., et al., Data for Biochemical Research, Oxford, Clarendon Press, 1959.
- ^ a b c Franz Dietrich Klingler; Wolfgang Ebertz (2005). "Oxocarboxylic Acids". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a18_313. ISBN 978-3527306732.
- ^ a b Lubert Stryer (1981). Biochemistry (2nd ed.).
- ^ Stryer, Lubert (1995). Biochemistry (Fourth ed.). New York: W.H. Freeman and Company. pp. 510–515, 581–613, 775–778. ISBN 0-7167-2009-4.
- ^ Robert C. Krueger (1952). "Crystalline Acetoacetic Acid". Journal of the American Chemical Society. 74 (21): 5536. doi:10.1021/ja01141a521.
- ^ Reynolds, George A.; VanAllan, J. A. (1952). "Methylglyoxal-ω-Phenylhydrazone". Organic Syntheses. 32: 84. doi:10.15227/orgsyn.032.0084; Collected Volumes, vol. 4, p. 633.
- ^ Hay, R. W.; Bond, M. A. (1967). "Kinetics of decarboxilation of acetoacetic acid". Aust. J. Chem. 20 (9): 1823–8. doi:10.1071/CH9671823.
- ^ Grande, Karen D.; Rosenfeld, Stuart M. (1980). "Tautomeric equilibriums in acetoacetic acid". The Journal of Organic Chemistry. 45 (9): 1626–1628. doi:10.1021/jo01297a017. ISSN 0022-3263.
- ^ Kiran Kumar Solingapuram Sai; Thomas M. Gilbert; Douglas A. Klumpp (2007). "Knorr Cyclizations and Distonic Superelectrophiles". J. Org. Chem. 72 (25): 9761–9764. doi:10.1021/jo7013092. PMID 17999519.
- ^ a b Nyenwe, EA; Kitabchi, AE (April 2016). "The evolution of diabetic ketoacidosis: An update of its etiology, pathogenesis and management". Metabolism: Clinical and Experimental. 65 (4): 507–21. doi:10.1016/j.metabol.2015.12.007. PMID 26975543.
- ^ Hartman, AL; Vining, EP (January 2007). "Clinical aspects of the ketogenic diet". Epilepsia. 48 (1): 31–42. doi:10.1111/j.1528-1167.2007.00914.x. PMID 17241206. S2CID 21195842.
- ^ Sumithran, Priya; Proietto, Joseph (2008). "Ketogenic diets for weight loss: A review of their principles, safety and efficacy". Obesity Research & Clinical Practice. 2 (1): I–II. doi:10.1016/j.orcp.2007.11.003. PMID 24351673.
- ^ Misra, S; Oliver, NS (28 October 2015). "Diabetic ketoacidosis in adults" (PDF). BMJ (Clinical Research Ed.). 351: h5660. doi:10.1136/bmj.h5660. hdl:10044/1/41091. PMID 26510442. S2CID 2819002.
- ^ Tatone, EH; Gordon, JL; Hubbs, J; LeBlanc, SJ; DeVries, TJ; Duffield, TF (1 August 2016). "A systematic review and meta-analysis of the diagnostic accuracy of point-of-care tests for the detection of hyperketonemia in dairy cows". Preventive Veterinary Medicine. 130: 18–32. doi:10.1016/j.prevetmed.2016.06.002. PMID 27435643.

