Histamine: Difference between revisions
→Sexual response: unverifiable |
Eric Kvaalen (talk | contribs) Started section on properties (modified from German Wikipedia). |
||
| Line 21: | Line 21: | ||
| MeltingPt= 83.5 °C (182.3 °F) |
| MeltingPt= 83.5 °C (182.3 °F) |
||
| BoilingPt= 209.5 °C (409.1 °F) |
| BoilingPt= 209.5 °C (409.1 °F) |
||
| pK<sub> |
| pK<sub>imidazole</sub> 6.04 |
||
| pK<sub>NH2</sub> 9.75 |
| pK<sub>NH2</sub> 9.75 |
||
| Solubility= |
| Solubility= |
||
| Line 32: | Line 32: | ||
}} |
}} |
||
'''Histamine''' is a [[biogenic amine]] involved in local [[immune system|immune responses]] as well as regulating physiological function in the gut and acting as a [[neurotransmitter]].<ref>{{cite book |author=Marieb, E. |title=Human anatomy & physiology |publisher=Benjamin Cummings |location=San Francisco |year=2001 |pages=414 |isbn=0-8053-4989-8 |oclc= |doi=}}</ref> It is found in virtually all animal body cells. New evidence also indicates that histamine plays an important role in [[chemotaxis]] of [[white blood cell]]s.{{Fact|date=November 2007}} |
'''Histamine''' is a [[biogenic amine]] involved in local [[immune system|immune responses]] as well as regulating physiological function in the gut and acting as a [[neurotransmitter]].<ref>{{cite book |author=Marieb, E. |title=Human anatomy & physiology |publisher=Benjamin Cummings |location=San Francisco |year=2001 |pages=414 |isbn=0-8053-4989-8 |oclc= |doi=}}</ref> It is found in virtually all animal body cells. New evidence also indicates that histamine plays an important role in [[chemotaxis]] of [[white blood cell]]s.{{Fact|date=November 2007}} |
||
==Properties== |
|||
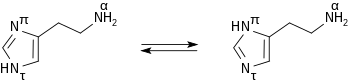
Histamine forms colorless [[hygroscopic]] [[crystal]]s that melt at 84°C, and are easily dissolved in [[water]] or [[ethanol]], but not in [[diethyl ether|ether]]. In [[aqueous solution]] histamine exists in two [[tautomer]]ic forms, ''N<sup>π</sup>-H''-histamine und ''N<sup>τ</sup>-H''-histamine. |
|||
{|align="left" |
|||
[[image:Histamine tautomers.svg|thumb|350px|Tautomers of histamine]] |
|||
|} |
|||
Histamine has two [[base (chemistry)|basic]] centres, namely the [[aliphatic]] [[amino group]] and whichever [[nitrogen]] atom of the [[imidazole]] ring does not already have a [[hydrogen|proton]]. Under physiological conditions, the aliphatic amino group (having a [[dissociation constant|pK]]<sub>a</sub> around 9.4) will be [[protonated]], whereas the second nitrogen of the imidazole ring (pK<sub>a</sub> ≈ 5.8) will not be protonated.<ref name="Paiva 1970">Paiva T.B., Tominaga M. & Paiva A.C. (1970). Ionization of histamine, N-acetylhistamine, and their iodinated derivatives. ''J. Med. Chem.'', '''13''', 689–692.</ref> |
|||
Thus, histamine is normally protonated to a singly-charged [[cation]]. |
|||
==Synthesis and metabolism== |
==Synthesis and metabolism== |
||
Revision as of 18:44, 29 August 2008
This article needs additional citations for verification. (January 2008) |
Template:Chembox new Histamine is a biogenic amine involved in local immune responses as well as regulating physiological function in the gut and acting as a neurotransmitter.[1] It is found in virtually all animal body cells. New evidence also indicates that histamine plays an important role in chemotaxis of white blood cells.[citation needed]
Properties
Histamine forms colorless hygroscopic crystals that melt at 84°C, and are easily dissolved in water or ethanol, but not in ether. In aqueous solution histamine exists in two tautomeric forms, Nπ-H-histamine und Nτ-H-histamine.
Histamine has two basic centres, namely the aliphatic amino group and whichever nitrogen atom of the imidazole ring does not already have a proton. Under physiological conditions, the aliphatic amino group (having a pKa around 9.4) will be protonated, whereas the second nitrogen of the imidazole ring (pKa ≈ 5.8) will not be protonated.[2] Thus, histamine is normally protonated to a singly-charged cation.
Synthesis and metabolism
Histamine is derived from the decarboxylation of the amino acid histidine, a reaction catalyzed by the enzyme L-histidine decarboxylase. It is a hydrophilic vasoactive amine.

Once formed, histamine is either stored or rapidly inactivated. Histamine released into the synapses is broken down by acetaldehyde dehydrogenase. It is the deficiency of this enzyme that triggers an allergic reaction as histamines pool in the synapses. Histamine is broken down by histamine-N-methyltransferase and diamine oxidase. Some forms of foodborne disease, so-called "food poisonings," are due to conversion of histidine into histamine in spoiled food, such as fish.
Storage and release

Most histamine in the body is generated in granules in mast cells (see figure) or in white blood cells called basophils. Mast cells are especially numerous at sites of potential injury - the nose, mouth, and feet; internal body surfaces; and blood vessels. Non-mast cell histamine is found in several tissues, including the brain, where it functions as a neurotransmitter. Another important site of histamine storage and release is the enterochromaffin-like (ECL) cell of the stomach.
The most important pathophysiologic mechanism of mast cell and basophil histamine release is immunologic. These cells, if sensitized by IgE antibodies attached to their membranes, degranulate when exposed to the appropriate antigen. Certain amines and alkaloids, including such drugs as morphine, and curare alkaloids, can displace histamine in granules and cause its release. Antibiotics like polymyxin are also found to be stimulating histamine release.
Mechanism of action
Histamine exerts its actions by combining with specific cellular histamine receptors. The four histamine receptors that have been discovered are designated H1 through H4.
| Typ | Standort | Function |
| H1 histamine receptor | Found on smooth muscle, endothelium, and central nervous system tissue | Causes vasodilation, bronchoconstriction, smooth muscle activation, separation of endothelial cells (responsible for hives), and pain and itching due to insect stings; the primary receptors involved in allergic rhinitis symptoms and motion sickness. |
| H2 histamine receptor | Located on parietal cells | Primarily stimulate gastric acid secretion |
| H3 histamine receptor | - | Decreased neurotransmitter release: histamine, acetylcholine, norepinephrine, serotonin |
| H4 histamine receptor | Found primarily in the thymus, small intestine, spleen, and colon. It is also found on basophils and in the bone marrow. | Unknown physiological role. |
Roles in the body
Sleep regulation
Histamine is released as a neurotransmitter. The cell bodies of neurons which release histamine are found in the posterior hypothalamus, in various tuberomammillary nuclei. From here, these histaminergic neurons project throughout the brain, to the cortex through the medial forebrain bundle. Histaminergic action is known to modulate sleep. Classically, antihistamines (H1 histamine receptor antagonists) produce sleep. Likewise, destruction of histamine releasing neurons, or inhibition of histamine synthesis leads to an inability to maintain vigilance. Finally, H3 receptor antagonists (which stimulate histamine release) increase wakefulness.
It has been shown that histaminergic cells have the most wakefulness-related firing pattern of any neuronal type thus far recorded. They fire rapidly during waking, fire more slowly during periods of relaxation/tiredness and completely stop firing during REM and NREM (non-REM) sleep. Histaminergic cells can be recorded firing just before an animal shows signs of waking.
Sexual response
Research has shown that histamine is released as part of the human orgasm from mast cells in the genitals. If this response is lacking this may be a sign of histapenia (histamine deficiency). In such cases, a doctor may prescribe diet supplements with folic acid and niacin (which used in conjunction can increase blood histamine levels and histamine release), or L-histidine.
Schizophrenia
It has been found that about half the patients classified as suffering from schizophrenia have low histamine levels in the blood.[3] This may be because of antipsychotics that have unwanted effect on histamine, such as quetiapine. Although, in these cases, as histamine levels were increased, their health improved.
Disorders
High or low histamine levels are considered by some of the alternative medicine community to be health issues; this is not accepted by the mainstream medical community.1 However, as an integral part of the immune system it may be involved in immune system disorders and allergies.
Nomenclature
"H substance" or "substance H" are occasionally used in medical literature for histamine or a hypothetical histamine-like diffusible substance released in allergic reactions of skin and in the responses of tissue to inflammation.
See also
References
Histamine and Histamine intolerance[1]
- ^ Marieb, E. (2001). Human anatomy & physiology. San Francisco: Benjamin Cummings. p. 414. ISBN 0-8053-4989-8.
- ^ Paiva T.B., Tominaga M. & Paiva A.C. (1970). Ionization of histamine, N-acetylhistamine, and their iodinated derivatives. J. Med. Chem., 13, 689–692.
- ^ What is Schizophrenia?
External links
