Boger pyridine synthesis: Difference between revisions
Appearance
Content deleted Content added
No edit summary |
added Category:Name reactions using HotCat |
||
| Line 17: | Line 17: | ||
[[Category:Organic reactions]] |
[[Category:Organic reactions]] |
||
[[Category:Name reactions]] |
|||
Revision as of 16:08, 30 October 2015
This article has multiple issues. Please help improve it or discuss these issues on the talk page. (Learn how and when to remove these template messages)
|
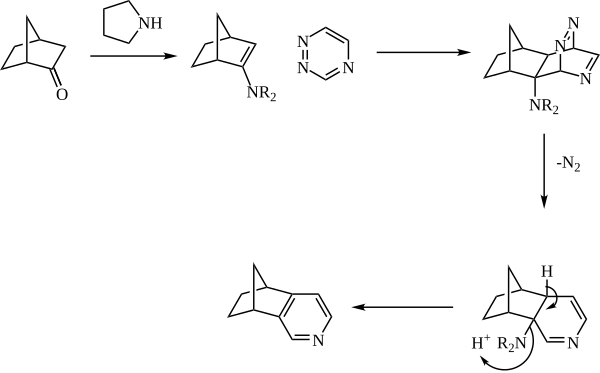
The Boger pyridine synthesis is a cycloaddition approach to the formation of pyridines in which an enamine reacts in an inverse-electron demand Diels-Alder reaction with a 1,2,4-triazine to form the pyridine nucleus. The reaction is especially useful for accessing pyridines that would be difficult or impossible to access via other methods.
Mechanism
The enamine is generally generated in situ from catalytic amine (such as pyrrolidine) and a ketone. The enamine then reacts as the dienophile with a 1,2,4-triazine. The initial adduct then expels nitrogen, and the pyridine is rearomatized with loss of the amine.
References
- Boger, D.; Panek, J.; Meier, M. (1982). "Diels-Alder reaction of heterocyclic azadienes". J. Org. Chem. 47: 895.
- Li, Jie Jack (2002). Name Reactions A Collection of Detailed Reaction Mechanisms. Berlin: Springer-Verlag. p. 40. ISBN 3-540-43024-5.
