Electrospun Biocomposite Polycaprolactone/Collagen Tubes as Scaffolds for Neural Stem Cell Differentiation
Abstract
:1. Introduction
2. Results and Discussion
2.1. Nanofiber Fabrication and Characterization
2.1.1. SEM Analysis

| Fiber diameter (nm) | Adhesion (%) | Proliferation (%) | Differentiation (% neurons) |
| 330 ± 17 | 92 | 84 | 38 |
| 510 ± 21 | 83 | 90 | 80 |
| 640 ± 83 | 68 | 81 | 58 |
| Fiber alignment | Adhesion (%) | Proliferation (%) | Differentiation (%) |
| Random | 73 | 52 | 56 |
| Aligned | 94 | 71 | 73 |
2.1.2. Tensile Strength

2.1.3. Degradation

2.2. Growth Factor Release

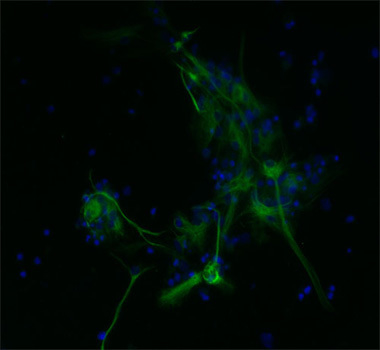
2.3. In vitro Differentiation and Immunocytochemisty



3. Experimental Section
3.1. Electrospinning of Nanofibers
3.2. Morphology and Characterization of Electrospun Nanofibers
3.3. Growth Factor Release
3.4. Tissue Preparation and Neural Stem Cell Cultures
3.5. In vitro Differentiation and Immunocytochemisty
3.6. Antibodies
3.7. Statistical Analysis
4. Conclusions
Acknowledgements
References and Notes
- Flax, J.D.; Aurora, S.; Yang, C.; Simonin, C.; Wills, A.M.; Billinghurst, L.L.; Jendoubi, M.; Sidman, R.L.; Wolfe, J.H.; Kim, S.U.; Snyder, E.Y. Engraftable human neural stem cells respond to developmental cues, replace neurons, and express foreign genes. Nat. Biotechnol. 1998, 16, 1033–1039. [Google Scholar] [CrossRef]
- Shihabuddin, L.S.; Horner, P.J.; Ray, J.; Gage, F.H. Adult spinal cord stem cells generate neurons after transplantation in the adult dentate gyrus. J. Neurosci. 2000, 20, 8727–8735. [Google Scholar] [PubMed]
- Oudega, M.; Xu, X.M. Schwann cell transplantation for repair of the adult spinal cord. J. Neurotrauma 2006, 23, 453–467. [Google Scholar] [CrossRef] [PubMed]
- Richardson, P.M.; McGuinness, U.M.; Aguayo, A.J. Axons from CNS neurons regenerate into PNS grafts. Nature 1980, 284, 264–265. [Google Scholar] [CrossRef] [PubMed]
- David, S.; Aguayo, A.J. Axonal elongation into peripheral nervous system "bridges" after central nervous system injury in adult rats. Science 1981, 214, 931–933. [Google Scholar] [CrossRef] [PubMed]
- Tsai, E.C.; Krassioukov, A.V.; Tator, C.H. Corticospinal regeneration into lumbar grey matter correlates with locomotor recovery after complete spinal cord transection and repair with peripheral nerve grafts, fibroblast growth factor 1, fibrin glue, and spinal fusion. J. Neuropathol. Exp. Neurol. 2005, 64, 230–244. [Google Scholar] [PubMed]
- Cheng, H.; Cao, Y.; Olson, L. Spinal cord repair in adult paraplegic rats: partial restoration of hind limb function. Science 1996, 273, 510–513. [Google Scholar] [CrossRef] [PubMed]
- Tsai, E.C.; Tator, C.H. Neuroprotection and regeneration strategies for spinal cord repair. Curr. Pharm. Des. 2005, 11, 1211–1122. [Google Scholar] [CrossRef] [PubMed]
- Tsai, E.C.; Dalton, P.D.; Shoichet, M.S.; Tator, C.H. Synthetic hydrogel guidance channels facilitate regeneration of adult rat brainstem motor axons after complete spinal cord transection. J. Neurotrauma 2004, 21, 789–804. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.M.; Guenard, V.; Kleitman, N.; Bunge, M.B. Axonal regeneration into Schwann cell-seeded guidance channels grafted into transected adult rat spinal cord. J. Comp. Neurol. 1995, 351, 145–60. [Google Scholar] [CrossRef] [PubMed]
- Spilker, M.H.; Yannas, I.V.; Hsu, H.P.; Norregaard, T.V.; Kostyk, S.K.; Spector, M. The effects of collagen-based implants on early healing of the adult rat spinal cord. Tissue Eng. 1997, 3, 309–317. [Google Scholar] [CrossRef]
- Oudega, M.; Gautier, S.E.; Chapon, P.; Fragoso, M.; Bates, M.L.; Parel, J.M.; Bunge, M.B. Axonal regeneration into Schwann cell grafts within resorbable poly(alpha-hydroxyacid) guidance channels in the adult rat spinal cord. Biomaterials 2001, 22, 1125–1136. [Google Scholar] [CrossRef] [PubMed]
- Shih, Y.R.V.; Chen, C.N.; Tsai, S.W.; Wang, Y.J.; Lee, O.K. Growth of mesenchymal stem cells on electrospun type I collagen nanofibers. Stem Cells 2006, 24, 2391–2397. [Google Scholar] [CrossRef] [PubMed]
- Prabhakaran, M.P.; Venugopal, J.; Chan, C.K.; Ramakrishna, S. Surface modified electrospun nanofibrous scaffolds for nerve tissue engineering. Nanotechnology 2008, 19, 455102–455109. [Google Scholar] [CrossRef] [PubMed]
- Venugopal, J.; Low, S.; Choon, A.T.; Kumar, A.B.; Ramakrishna, S. Electrospun-modified nanofibrous scaffolds for the mineralization of osteoblast cells. J. Biomed. Mater. Res. Part A 2008, 85, 408–417. [Google Scholar] [CrossRef]
- Sabine, W.G.; Hans, G.; Leprince, P.; Rigo, J.M.; Gustave, M.; Rogister, B. Plasticity of cultured mesenchymal stem cells: switch from nestin-positive to excitable neuron-like phenotype. Stem Cells 2005, 23, 392–402. [Google Scholar] [CrossRef] [PubMed]
- Lemmouchi, Y.; Schacht, E. Preparation and in vitro evaluation of biodegradable poly (3-caprolactone-co-D,L lactide) (X–Y) devices containing tryparocidal drugs. J. Control. Release 1997, 45, 227–233. [Google Scholar] [CrossRef]
- Ye, W.P.; Du, F.S.; Jin, W.H.; Yan, J.Y.; Xu, Y. In vitro degradation of poly(caprolactone), poly(lactide) and their block copolymers influence of composition temperature of composition temperature and morphology. React. Funct. Polym. 1997, 32, 161–168. [Google Scholar] [CrossRef]
- Lucchesi, C.; Barbanti, S.H.; Joazeiro, P.P.; Duek, E.A. Cell culture on PCL/PLGA blends. J. Appl. Polym. Sci. 2009, 115, 2609–2615. [Google Scholar] [CrossRef]
- Sasmazel, H.T.; Gumusderelioglu, M.; Gurpinar, A.; Onur, M.A. Comparison of cellular proliferation on dense and porous PCL scaffolds. Bio-Med. Mat. Eng. 2008, 18, 119–128. [Google Scholar]
- Place, E.S.; George, J.H.; Williams, C.K.; Stevens, M.M. Synthetic polymer scaffolds for tissue engineering. Chem. Soc. Rev. 2009, 38, 1139–1151. [Google Scholar] [CrossRef] [PubMed]
- Okano, H. Neural stem cells as therapeutic agents for CNS injuries and disorders. Int. Congr. Ser. 2003, 1252, 493–498. [Google Scholar] [CrossRef]
- Chojnacki, A.; Weiss, S. Pigment epithelium-derived growth factor: modulating adult neural stem cell self-renewal. Nat. Neurosci. 2009, 12, 1481–1483. [Google Scholar] [CrossRef] [PubMed]
- Christopherson, G.T.; Song, H.; Mao, H.Q. The influence of fiber diameter of electrospun substances on stem cell differentiation and proliferation. Biomaterials 2009, 30, 556–564. [Google Scholar] [CrossRef] [PubMed]
- Ma, M.; Wei, P.; Wei, T.; Ransohoff, R.M.; Jakeman, L.B. Enhanced axonal growth into a spinal cord contusion injury site in a strain of mouse (129X1/SvJ) with a diminished inflammatory response. J. Comp. Neurol. 2004, 474, 469–486. [Google Scholar] [CrossRef] [PubMed]
- Borschel, G.H.; Kia, K.F.; Kuzon, W.M.; Dennis, R.G. Mechanical properties of acellular peripheral nerve. J. Surg. Res. 2003, 114, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Richardson, S.M. Tissue engineering today, not tomorrow. Regen. Med. 2007, 2, 91–94. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, S.; Ang, L.T.; Goh, J.C.H.; Toh, S.L. Growth factor delivery through electrospun nanofibers in scaffolds for tissue engineering applications. J. Biomed. Mater. Res. Part A 2009, 4, 1539–1550. [Google Scholar]
- Wells, M.R; Kraus, K.; Batter, D.K.; Blunt, D.G.; Weremowitz, J.; Lynch, S.E.; Antoniades, H.N.; Hansson, H.A. Gel matrix vehicles for growth factor application in nerve gap injuries repaired with tubes: a comparison of biomatrix, collagen, and methylcellulose. Exp.Neurol. 1997, 146, 395–402. [Google Scholar] [CrossRef]
- Cheng, H.; Liao, K.K.; Liao, S.F.; Chuang, T.Y.; Shih, Y.H. Spinal cord repair with acidic fibroblast growth factor as a treatment for a patient with chronic paraplegia. Spine 2004, 29, E284–E288. [Google Scholar] [CrossRef] [PubMed]
- Tuszynski, M.H.; Peterson, D.A.; Ray, J.; Baird, A.; Nakahara, Y.; Gage, F.H. Fibroblasts genetically modified to produce nerve growth factor induce robust neuritic ingrowth after grafting to the spinal cord. Exp. Neurol. 1994, 126, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Tsai, E.C.; Dalton, P.D.; Shoichet, M.S.; Tator, C.H. Matrix inclusion within synthetic hydrogel guidance channels improves specific supraspinal and local axonal regeneration after complete spinal cord transection. Biomaterials 2006, 27, 519–533. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.Y.; Peters, M.C.; Anderson, K.W.; Mooney, D.J. Controlled growth factor release from synthetic extracellular matrices. Nature 2000, 408, 998–1000. [Google Scholar] [CrossRef] [PubMed]
- Svendsen, C.N.; Caldwell, M.A.; Shen, J.; ter Borg, M.G.; Rosser, A.E.; Tyers, P.; Karmiol, S.; Dunnett, S.B. Long-Term Survival of Human Central Nervous System Progenitor Cells Transplanted into a Rat Model of Parkinson's Disease. Exp. Neurol. 1997, 148, 135–146. [Google Scholar] [CrossRef] [PubMed]
- McBride, J.L.; Behrstock, S.P.; Chen, E.Y.; Jakel, R.J.; Siegel, I.; Svendsen, C.N.; Kordower, J.H. Human neural stem cell transplants improve motor function in a rat model of Huntington's disease. J. Comp. Neurol. 2004, 475, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Wislet-Gendebien, S.; Leprince, P.; Moonen, G.; Rogister, B. Regulation of neural markers nestin and GFAP expression by cultivated bone marrow stromal cells. J. Cell Sci. 2003, 116, 3295–3302. [Google Scholar] [CrossRef] [PubMed]
- Li, X.K.; Guo, A.C.; Zuo, P.P. Survival and differentiation of transplanted neural stem cells in mice brain with MPTP-induced Parkinson disease. Acta Pharmacol. Sin. 2003, 24, 1192–1198. [Google Scholar] [PubMed]
- Badami, A.S.; Kreke, M.R.; Thompson, M.S.; Riffle, J.S.; Goldstein, A.S. Effect of fiber diameter on spreading, proliferation, and differentiation of osteoblastic cells on electrospun poly(lactic acid) substrates. Biomaterials 2006, 27, 596–606. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Murugan, R.; Wang, S.; Ramakrishna, S. Electrospinning of nano/micro scale poly(L-lactic acid) aligned fibers and their potential in neural tissue engineering. Biomaterials 2005, 26, 2603–2610. [Google Scholar] [CrossRef] [PubMed]
- Rosso, F.; Giordano, A.; Barbarisi, M.; Barbarisi, A. From cell-ECM interactions to tissue engineering. J. Cell. Physiol. 2004, 199, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Mobarakeh, L.G.; Prabhakaran, M.P.; Morshed, M.; Esfahani, M.H.N.; Ramakrishna, S. Electropsun PCL/gelatin nanofibrous scaffolds for nerve tissue engineering. Biomaterials 2008, 29, 4532–4539. [Google Scholar] [PubMed]
- Liu, Y.; Gan, L.; Carlsson, D.J.; Fagerholm, P.; Lagali, N.; Watsky, M.A.; Munger, R.; Hodge, W.G.; Priest, D.; Griffith, M. A simple, cross-linked collagen tissue substitute for corneal implantation. Invest. Ophthalmol. Vis. Sci. 2006, 47, 1869–1875. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, B.A.; Weiss, S. Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science 1992, 255, 1707–1710. [Google Scholar] [CrossRef] [PubMed]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an Open Access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hackett, J.M.; Dang, T.T.; Tsai, E.C.; Cao, X. Electrospun Biocomposite Polycaprolactone/Collagen Tubes as Scaffolds for Neural Stem Cell Differentiation. Materials 2010, 3, 3714-3728. https://doi.org/10.3390/ma3063714
Hackett JM, Dang TT, Tsai EC, Cao X. Electrospun Biocomposite Polycaprolactone/Collagen Tubes as Scaffolds for Neural Stem Cell Differentiation. Materials. 2010; 3(6):3714-3728. https://doi.org/10.3390/ma3063714
Chicago/Turabian StyleHackett, Joanne M., ThucNhi T. Dang, Eve C. Tsai, and Xudong Cao. 2010. "Electrospun Biocomposite Polycaprolactone/Collagen Tubes as Scaffolds for Neural Stem Cell Differentiation" Materials 3, no. 6: 3714-3728. https://doi.org/10.3390/ma3063714