Lanthanum phosphide
| |
| Names | |
|---|---|
| Other names
Phosphanylidynelanthanum
| |
| Identifiers | |
3D model (JSmol)
|
|
| ECHA InfoCard | 100.042.515 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| LaP | |
| Molar mass | 169.88 |
| Appearance | Black crystals |
| Density | 5.2 g/cm3 |
| Reacts with water | |
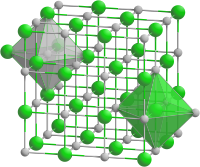
| Structure[1] | |
| Rock salt structure | |
| Fm3m | |
a = 0.6025 nm
| |
Formula units (Z)
|
4 |
| Octahedral at La3+, Octahedral at P3- | |
| Related compounds | |
Other anions
|
Lanthanum nitride Lanthanum arsenide Lanthanum bismuthide |
Other cations
|
Scandium phosphide Yttrium phosphide Cerium phosphide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Lanthanum phosphide is an inorganic compound of lanthanum and phosphorus with the chemical formula LaP.
Synthesis
[edit]Lanthanum phosphide can be made by heating lanthanum metal with excess phosphorus in a vacuum:[2]
- 4 La + P4 → 4 LaP
Physical properties
[edit]Lanthanum phosphide forms black crystals of a cubic system, space group Fm3m, cell parameters a = 0.6025 nm, with number of formulas per unit cell Z = 4.[1]
The crystals are very unstable and decompose in the open air.
Electronic properties
[edit]Lanthanum phosphide is an example of a strongly correlated material,[3] complicating theoretical prediction of its properties.
According to HSE06 calculations, lanthanum phosphide has been theoretically predicted to have an indirect band gap of 0.25 eV along the Γ-X direction.[4] According to HSE06 calculations with spin-orbit coupling, the band gap is predicted to be a direct gap of 0.72 eV at the X point.[5] Using EVGGA, the compound is predicted to have a band gap of 0.56 eV along the Γ-X direction.[6] FP-LAPW has predicted an indirect gap of 0.33 eV along the Γ-X direction.[3]
Chemical properties
[edit]Lanthanum phosphide reacts with water, releasing highly toxic phosphine gas:
- LaP + 3H2O → La(OH)3 + PH3
Uses
[edit]Lanthanum phosphide compound is a semiconductor used in high power, high frequency applications, and in laser diodes.[7][8]
Lanthanum polyphosphide
[edit]In addition to the simple phosphide, LaP, lanthanum and phosphorus can also form phosphorus-rich compounds such as LaP2[9] LaP5[10] and LaP7.[11]
References
[edit]- ^ a b Standard X-ray Diffraction Powder Patterns (PDF). U.S. Department of Commerce, National Bureau of Standards. 1967-08-31. p. 69. Retrieved 2023-11-04.
- ^ Samsonov, G.V.; Endrzheevskaya, S.N. (September 1963). "Production and Some Properties of Lanthanum Phosphide". Journal of General Chemistry of the USSR (English Translation). 33 (9). Consultants Bureau: 2729. Google books Journal index
- ^ a b Shoaib, M.; Murtaza, G.; Khenata, R.; Farooq, M.; Ali, Roshan (2013). "Structural, elastic, electronic and chemical bonding properties of AB (A=Sc,Y,La;B=N,P,As,Sb,Bi) from first principles". Computational Materials Science. 79. Elsevier BV: 239–246. doi:10.1016/j.commatsci.2013.06.015. ISSN 0927-0256.
- ^ Yan, X. Z.; Chen, Y. M.; Kuang, X. Y.; Xiang, S. K. (2014-08-26). "Theoretical investigation of La monopnictides: Electronic properties and pressure-induced phase transition". Journal of Applied Physics. 116 (8). AIP Publishing: 083707. Bibcode:2014JAP...116h3707Y. doi:10.1063/1.4893645. ISSN 0021-8979.
- ^ Zhou, Yu; Tao, Wang-Li; Zeng, Zhao-Yi; Chen, Xiang-Rong; Chen, Qi-Feng (2019-01-28). "Thermoelectric properties of topological insulator lanthanum phosphide via first-principles study". Journal of Applied Physics. 125 (4). AIP Publishing: 045107. Bibcode:2019JAP...125d5107Z. doi:10.1063/1.5043170. ISSN 0021-8979.
- ^ Charifi, Z.; Reshak, Ali Hussain; Baaziz, H. (2008). "Phase transition of LaX (X = P, As, Sb and Bi) at high pressure: Theoretical investigation of the structural and electronic properties". Solid State Communications. 148 (3–4). Elsevier BV: 139–144. Bibcode:2008SSCom.148..139C. doi:10.1016/j.ssc.2008.07.038. ISSN 0038-1098.
- ^ Lewis, Robert A. (30 March 2016). Hawley's Condensed Chemical Dictionary. John Wiley & Sons. ISBN 978-1-119-19372-2. Retrieved 21 December 2021.
- ^ O'Bannon, Loran (6 December 2012). Dictionary of Ceramic Science and Engineering. Springer Science & Business Media. p. 151. ISBN 978-1-4613-2655-7. Retrieved 21 December 2021.
- ^ v. Schnering, H. G.; Wichelhaus, W.; Nahrup, M. Schulze (1975). "Zur Chemie und Strukturchemie der Phosphide und Polyphosphide. XI. Lanthandiphosphid LaP2; Darstellung, Struktur und Eigenschaften". Zeitschrift für anorganische und allgemeine Chemie. 412 (3). Wiley: 193–201. doi:10.1002/zaac.19754120302. ISSN 0044-2313.
- ^ Wichelhaus, W.; Schnering, H. (1976). "Zur Chemie und Strukturchemie der Phosphide und Polyphosphide. 12. Die Pentaphosphide des Lanthans und Neodyms, LaP5 und NdP5". Zeitschrift für Anorganische und Allgemeine Chemie. 419: 77–86. doi:10.1002/ZAAC.19764190113. S2CID 97289383.
- ^ Raghavan, V. (2004). "Fe-La-P (iron-lanthanum-phosphorus)". Journal of Phase Equilibria and Diffusion. 25 (2). Springer Science and Business Media LLC: 172–173. doi:10.1007/s11669-004-0019-7. ISSN 1547-7037.
Further reading
[edit]- Iandelli, A.; Botti, R. (1936). "On the crystal structure of compounds of the rare earths with metalloids of the V group. Phosphides of La, Ce, Pr". Rendiconti Lincei. VI. 24. Accademia dei Lincei: 459. ISSN 0001-4435. OCLC 8450083. HathiTrust (no access)
