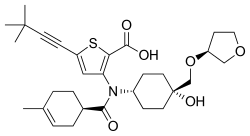
Radalbuvir
Appearance
 | |
| Clinical data | |
|---|---|
| Other names | GS-9669 |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C30H41NO6S |
| Molar mass | 543.72 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Radalbuvir (INN,[1] also known as GS-9669) is an experimental antiviral drug for the treatment of hepatitis C virus (HCV) infection developed by Gilead Sciences. Radalbuvir acts as an NS5B inhibitor. It is currently in clinical trials.[2] It targets NS5B polymerase.[3]
References
[edit]- ^ "International Nonproprietary Names for Pharmaceutical Substances" (PDF). WHO Drug Information. Vol. 28, no. 4. 2014. Archived from the original (PDF) on 2015-04-02.
- ^ "Search of: GS-9669 — List Results". ClinicalTrials.gov.
- ^ Borgia G, Maraolo AE, Nappa S, Gentile I, Buonomo AR (March 2018). "NS5B polymerase inhibitors in phase II clinical trials for HCV infection". Expert Opinion on Investigational Drugs. 27 (3): 243–250. doi:10.1080/13543784.2018.1420780. PMID 29271672. S2CID 3672885.
